A Bacteria That Destroy Plastics: A Sustainable Solution to PET Pollution

Plastic disposal has become a global environment concern. Worldwide eenvironmentalists and scientists are making efforts to seek effective solution of the growing accumulation of plastic and microplastic contamination in water bodies, landfills, air, and even food sources. To permanently deal with the growing plastic hazards, microbiologists are exploring the vast and diverse world of microbes as a reliable and promising solutions for plastic degradation. Recent advances have been made to use different microbial strains to degrade various type of plastics such as polyethylene (PE), polystyrene (PS), and polyethylene terephthalate (PET). PET is commonly used in beverage bottles, packaging containers, and synthetic fibres.
Bacteria that destroy plastic
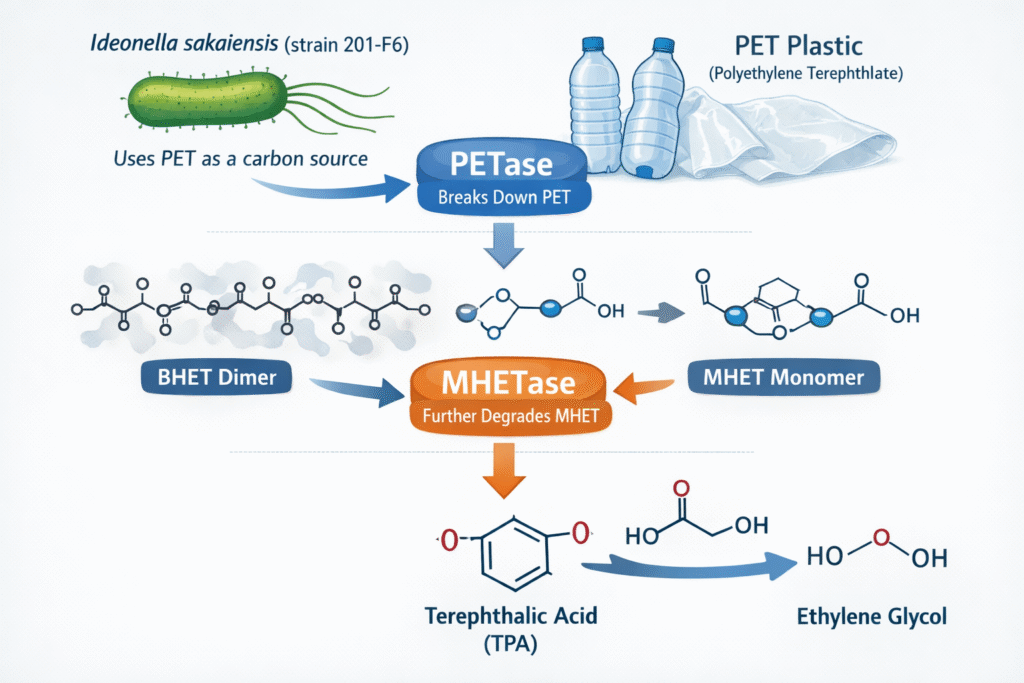
Ideonella sakaiensis (strain 201-F6), is known since decades as one of the most efficient PET degraders in the environment. This strain produces enzymes PETase and MHETase that hydrolyse PET and its resultant intermediate mono(2-hydroxyethyl) terephthalic acid into terephthalic acid (TPA) and ethylene glycol. PETase is an esterase that triggers the breakdown of ester bonds of PET in plastic polymer, degrading it to monomer MHET and dimer BHET. The resultant MHET is further broken down into TPA and ethylene glycol by the MHETase.
Green algae species Chlamydomonas reinhardtii (strain CC-124) has been also found the similar PET degrading potential. However, studies show that the degrading efficiency of these microbes largely depends upon the geometric property of the polymer, as larger surface area is generally degraded more quickly than those having smaller surfaces. Other contributing factors involve moisture, pH, aerobic environment, polymer melting point, additives, and biosurfactants. Other enzymes of primary importance in the process of plastic decomposition include esterases, laccase and cutinase that are present in bacteria and fungi, such as Candida antarctica, Thermobifida fusca and Aspergillus species. Cutinase enzymes are primarily effective in biodegradation of hydrophobic polymers while laccases, produced by Trametes versicolor and Pleurotus ostreatus, oxidises and degrades the plastic polymers by losing electrons and oxidizing the molecular oxygen, thus breaking the polymer chains of aromatic rings.
Study Outcomes:
Scientists at the Department of Applied Biology, Japan, discovered a novel bacterium, Ideonella sakaiensis 201-F6, that is able to use PET as its major energy and carbon source.
- 250 PET debris was collected from the contaminated environmental samples including sediment, soil, wastewater, and activated sludge from a PET bottle recycling site.
- Microbes from these samples were screened for their efficiency to use low-crystallinity (1.9%) PET film as the major carbon source for growth, in the modified lettuce and egg medium that was changed biweekly.
- After 20 days, one of the microbial consortium was found to degrade the PET film surface at a rate of 0.13mgcm–2 day–1at 30°C and 75% of the degraded PET film carbon was catabolized into CO2 at 28°C.
- Limiting dilutions of this consortium sample helped in isolation of a microbial strain, named Ideonella sakaiensis 201-F6.
- The strain was further cultured in the YSV (yeast extract–sodium carbonate vitamins) medium on the PET film and SEM images taken for 60 hours. The microbial cells of Ideonella sakaiensis were seen to be connected to the film and to each other by appendages., that might assist the delivery of secreted enzymes into the film.
- After 6 weeks, the PET film was damaged extensively at 30°C.
- The subconsortium that lacked the PET degrading capability lacked Ideonella sakaiensis, indicating its prime functionality in PET degradation.
- Further, genome sequence of Ideonella sakaiensis, identified one open reading frame (ORF), ISF6_4831, that encodes a putative lipase that shares 51% amino acid sequence identity with the catalytic residues of the hydrolase from mobifida fusca (TfH), known for its PET hydrolytic activity.
- The bacterial genes were expressed recombinantly and incubated with the PET film at 30°C for 18hours that led to the development of prominent pitting on the film surface.
- HPLC evaluation of the products released from the PET film revealed the presence of MHET together with the minor amounts of TPA and bis(2-hydroxyethyl) TPA(BHET), hydrolysed by the ISF6_4831 proteins.
- PET hydrolyzing activities of ISF6_4831 proteins, termed PETase, was found to be 120, 5.5, and 88 times higher than TfH (thermophilic actinomycete), cutinase from homolog from leaf-branch compost metagenome (LCC) and from F. solani (FsC) while its p-nitrophenol–linked aliphatic esters (pNP-aliphatic esters) degrading activity was found to be lower.
- ISF6_4831 proteins were found to prefer aliphatic esters that can degrade highly crystallized commercial bottle–derived PET.
- PETase is a heat labile enzyme that is more active against PET film at low temperature, capable of hydrolyzing PET with less enzyme diffusion into the aqueous phase and/or plastic vessels used for the reaction.
- RNA sequencing of the sakaiensis when grown on the PET film, revealed that the transcript level of PETase encoding gene was 15, 31, and 41 times highest among all analysed coding sequences, as compared to when bacteria was grown on maltose, TPA-Na and BHET, respectively.
- Another ISF6_0224 protein, designated as MHET hydrolase was found to be responsible for specific conversion of MHET to TPA and EG in sakaiensis, with a turn overrate (kcat) of 31±0.8s−1 and a Michaelis constant (Km)of 7.3±0.6mM.
Future Applications:
The substrate specificity and prominent hydrolytic activity of PETase, secreted by I.sakaiensi, is a critical finding that could revolutionalize the degradation and treatment technologies of the PET plastics in most environments. More efficient biotechnological recycling systems and enzyme-based plastic treatment technologies could be developed where plastic-degrading microbes or their enzymes are used to convert plastic waste into reusable monomers. These monomers, such as terephthalic acid and ethylene glycol from PET, can be repurposed for the synthesis of new plastics, supporting a circular economy. Further, microbial stabilizing synthetic consortia could be developed that combine bacteria, fungi, and actinomycetes to target different stages of polymer breakdown and mineralization. Such consortia could be applied in controlled composting systems, landfills, or wastewater treatment plants.