Preclinical Screening of T-Cell Therapies
T-cell therapy, has evolved as personalized immunotherapy that engineer patient’s T-cells receptors (TCR) to specifically identify the target cancer cells, autoimmune cells and other chronic infectious viruses. When these T cells bind to the targeted malignant antigen, the downstream signals within them get activated leading to targeted cell killing. These genetically engineered T-cells have enhanced affinity for specific tumor peptide-MHC complexes While this improves therapeutic potency, it also increases the risk of weak TCR cross-reactivity with antigens expressed on healthy tissues, potentially resulting in serious clinical toxicities. In the pre-clinical settings, interspecies differences in proteomic composition reduces the predictive value of in vivo toxicological models for assessing on-target and off-target TCR toxicity.
To mitigate these risks, Sanderson and colleagues at the (Adaptimmune (ADAPY) developed an extensive in vitro pre-clinical testing protocol to evaluate the safety and efficacy of T-cell therapies like CAR-T and specific peptide enhanced affinity receptor (SPEAR) T cells. The strategy involves potency testing of engineered T-cells using 2D and 3D human cell cultures and primary tumor material, in addition to safety testing using human primary cell and cell-line cross reactivity screening and molecular analysis to predict peptides recognized by the affinity-enhanced TCR.
Pre-clinical Screening of T-cell Therapy
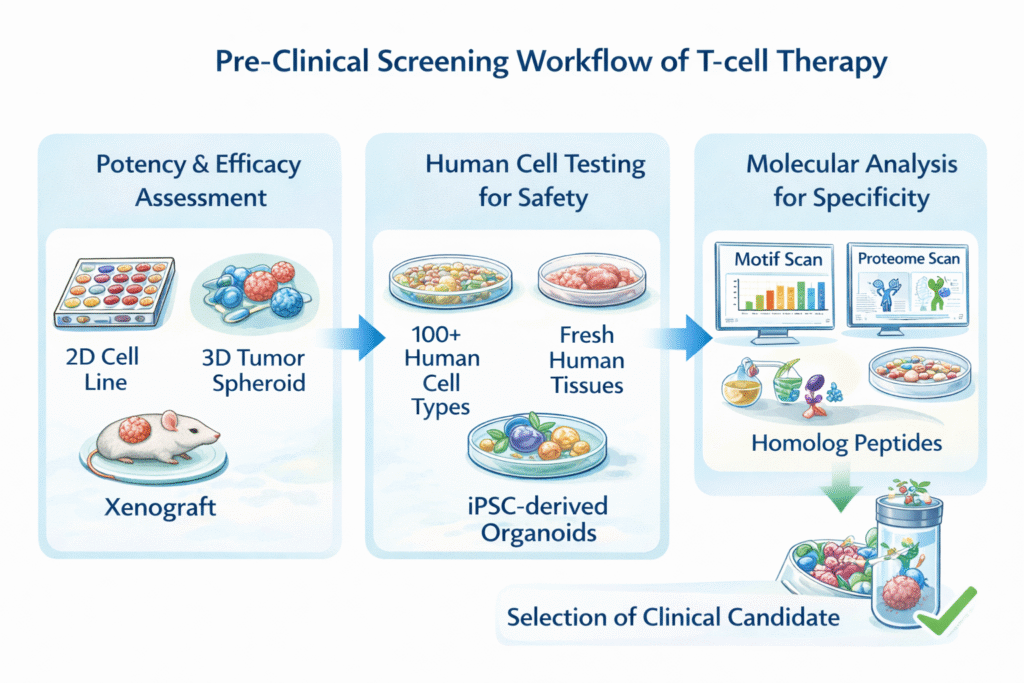
This approach follows 3 steps guided by screening and evaluation of T-cell therapy
Step 1: Potency & Efficacy Assessment
The process begins by confirming the anti-tumor activity of engineered T-cell therapy.
- 2D cell line assays are used to measure cytokine release and cytotoxicity.
- Antigen-driven proliferation assays assess T-cell expansion upon target recognition.
- Primary tumor samples are tested to confirm activity against clinically relevant material.
- In vivo efficacy studies evaluate tumor control in xenograft models.
- 3D cell-line models provide a more physiologically relevant assessment of tumor killing.
Only T-cell therapy that show strong and consistent efficacy progress further.
Step 2: Human Cell Testing for Safety
At this step, on-target and off-target toxicities are assessed using human-relevant systems.
- A standard human cell screen covering more than 100 normal cell types is performed to detect unintended reactivity.
- Fresh human tissues, organotypic cultures, and iPSC-derived models are used to improve biological relevance.
- Alloreactivity assays test whether the TCR reacts with alternative HLA alleles, identifying patient populations that must be excluded.
This step filters out T-cell therapy with unacceptable safety risks.
Step 3: Molecular Analysis for Specificity
Molecular studies are then used to understand and predict peptide cross-reactivity of TCRs.
- An X-scan systematically mutates each position of the target peptide to define tolerated substitutions.
- In silico searches use the resulting motif to scan the human proteome for potential mimetic peptides.
- Homolog identification examines related proteins, particularly within antigen families.
- Peptide screening tests candidate peptides for functional T-cell activation.
- Overexpression studies confirm whether candidate off-target peptides are naturally processed and presented.
This step provides mechanistic confidence that there is no clinically relevant off-target recognition is likely.
Step 4: Selection of a Clinical Candidate
Only T-cell therapy that demonstrate:
- Robust anti-tumor efficacy
- Minimal off-target and alloreactive responses
- No functionally relevant molecular cross-reactivity
are advanced to clinical trial as a potent anti-cancer candidate.
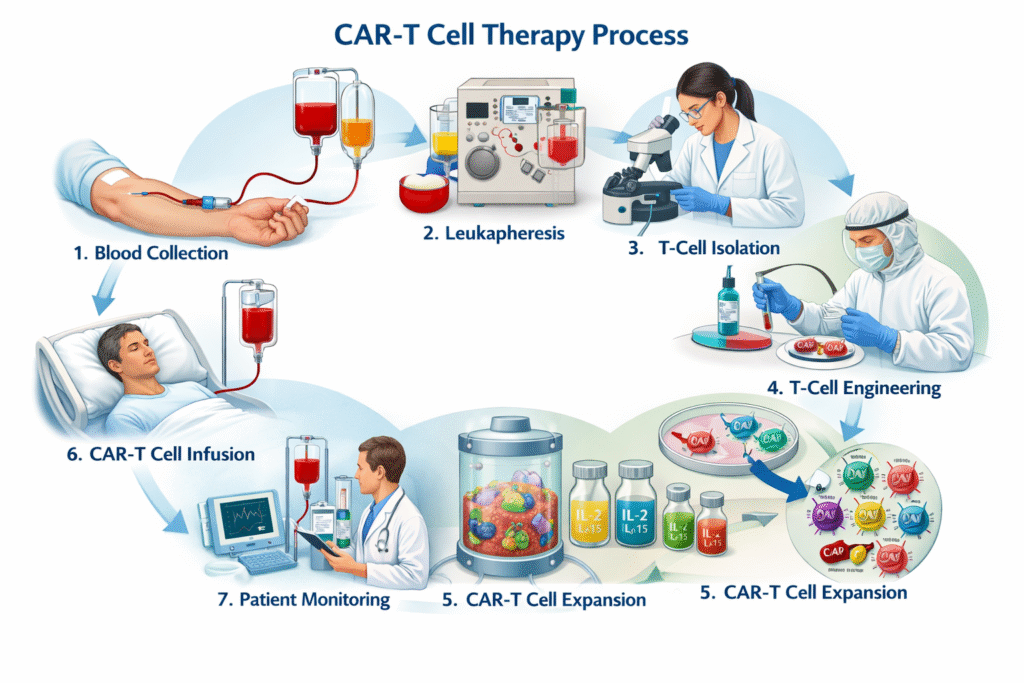
CAR-T Cell Therapy Process
In the clinical settings the therapy is conducted in a controlled environment by the well trained and proficient group of scientists and doctors.
The whole process of CAR-T cell therapy lasts for approximately 4-6 weeks
- Collection of Blood: At first, patients’ blood is collected using hypodermic needle attached to a Vacutainer or a syringe.
- Collection of White Blood Cells (Leukapheresis): The collected blood is then separated into white blood cells, red blood cells and plasma using density gradient centrifugation. The white blood cells are retained while the other components are returned to the patient. Ficoll density gradient is used to further retain the peripheral blood mononuclear cells (PBMCs).
- Isolation of T-cells: Targeted antibodies that binds to the T-cell surface markers are used to isolate the T-cells using various techniques like Magnetic-Activated Cell Sorting (MACS), Fluorescence-Activated Cell Sorting (FACS), Microbubble Technology, Microfluidics.
- T-cell Engineering: T-cells are modified with chimeric antigen receptor (CARs) to identify the specific targeted cancer markers. These T-cells simultaneously recognize tumor cells and get activated, creating CAR-T cells.
- CAR-T cell culture: CAR-T cells are cultured ex vivo and stimulated by cytokines like IL-2, IL-7, IL-15, and IL-21 to produce its multiple copies. These cells are also frozen for future administration.
- Infusion of CAR-T cells: The proliferated CAR-T cells, engineered to recognize the specific cancer markers, are transfused back into the patients at an appropriate dose.
- Patient’s Monitoring: Patients subjected to CAR-T cel therapy are monitored closely to control severe physical reactions, if any, in the following days.