Drug VV116: New Hope in the Fight Against Nipah Virus
Nipah virus encephalitis is described as acute febrile illness or brain inflammation, caused by Nipah virus. Nipah virus as single-stranded, negative-sense RNA virus. This virus belongs to the Henipavirus genus within the family Paramyx-oviridae, recognised as highly pathogenic zoonotic RNA viruses, carried by Pteropus bats (fruit bats), while pigs and horses may be the intermediate hosts.
After its first detection in Malaysia in 1999, the virus caused a major outbreak in Kerala in May 2018, killing over 17 people in 7 days. After first outbreak in India, WHO recognised Nipah virus as a highest priority regional threat. Recently, the disease has re-emerged in West Bengal among the healthcare workers, causing 5 deaths. Common symptoms observed in these patients include high fever, dizziness, confusion, and acute encephalitis, leading to a mortality rate of around 40–75%.
Drug against Nipah Virus
Since 2018, scientists have been rigorously aiming to find cure of the disease and decrease the mortality rate caused by the virus. One such lab, at Wuhan Institute of Virology, China, has found a potential anti-viral drug VV116, as a potential candidate for treating Nipah virus infection. VV116 has been previously reported for its anti-viral potency against SARS-CoV-2 (COVID-19 virus). It is an oral nucleoside prodrug that has been approved in China and Uzbekistan, for the treatment of COVID-19.
When assessed in vitro, VV116 selectively acted against Nipah virus and its inhibiting activity was almost twice than Remdesivir, a COVID-19 medicine. In Phase I clinical trial, serum concentration of VV116 reached much higher following a single dose of 800 mg, enough to inhibit the virus growth.
In vivo experiments also showed that the VV116 effectively inhibited the Nipah Virus-B strain at a single dose of 400 mg/kg when administered orally. The drug was found to rapidly absorb within 1 hour with effective tissue penetration. The brain concentration of the drug was 20 folds lower than in lungs or spleen but sufficiently effective to supress the viral infection. In hamsters, the drug was successful in inhibiting the responses like thickened alveolar septa, infiltration of inflammatory cells and significant inflammatory reactions in blood vessels, achieving the survival rate of 67% with delayed signs of the disease.
Detailed research:
- Vero E6, kidney epithelial cells were used as host to induce Nipah viral infection to study the in vitro antiviral activity of VV116 against Nipah virus (NiV-M strain).
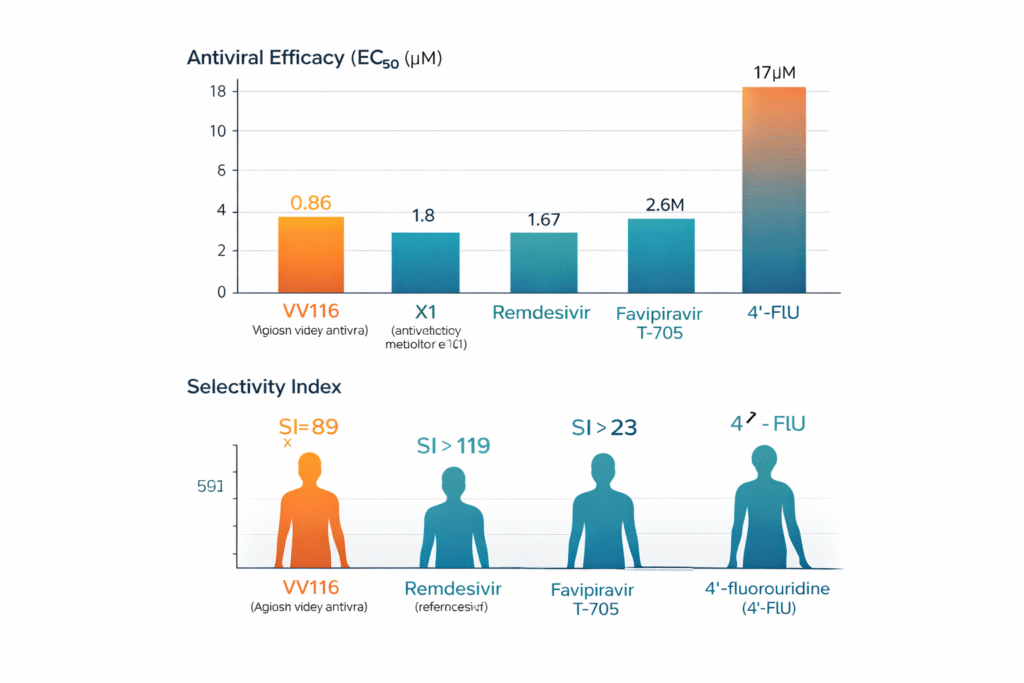
- Antiviral efficacy was measured by calculating the EC50 for VV116, X1 (active metabolite of VV116), Remdesivir ((reference antiviral drug), Favipiravir (T-705) (positive control antiviral), 4’-fluorouridine (4’-FlU) (control antiviral). The results found X1 (VV116) more potent than the other counterparts with the EC50 of 86 µM, 1.8 µM, 1.67 µM, 17 µM and 2.6 µM respectively.
- Cell viability was also assessed and Selectivity Index (SI) was calculated for these drugs was found to be (SI)= 89 for VV116 while SI >119, >23, 1>50 for Remdesivir, T-705 and 4’-fluorouridine.
- To assess the pharmacokinetic profile of VV116, golden Syrian hamsters were chosen and administered with the drug at doses of 200 mg/kg and 400 mg/kg. Then, Maximum plasma concentration (Cmax), Half-life (T1/2) and Area under the curve (AUC) was measured for X1. The drug showed linear PK, high plasma exposure and effective tissue penetration, suitable for in vivo antiviral testing.
- To assess the in vivo antiviral efficacy of the drug, hamsters were infected intraperitoneally with Nipah virus strain B at 1000 LD50. Post-infection, they were administered with VV116 at 200 mg/kg and 400 mg/kg, T-205 as positive control and a vehicle control, once daily for 14 days.
- After treatment, on day 4 lung, spleen and brain tissues were collected and viral RNA load was quantified using molecular methods like RT-PCR, showing dose-dependent reduction in viral loads in V116 group.
- Immunofluorescence staining on these tissue sample further validated reduced viral presence in V116 (400mg/kg) group. Also, lung tissues were found to be near- normal in this group while the vehicle control showed severe inflammation and lesions.
- Animal were also monitored for 21 days to assess the long-term survival after drug treatment. Parameters like body weight, Food/water intake, Disease symptoms and survival was recorded. In vehicle group there was 0% survival at day 13 while for VV116 there was 67% survival with maintained body weight.
Scope & Applications:
The study highlights the anti-NiV activity of VV116 and its metabolite X1, proposing it as a promising oral candidate against NiV infection. Further investigations may elucidate its inhibitory mechanism, particularly its potential activity against the viral RNA-dependent RNA polymerase, thus strengthening the current findings.
The scientist also suggests, VV116 could be safely used by humans at the dosage regimen of 800 mg twice a day and above to effectively inhibit the viral infection, providing strong recommendations for future NiV outbreaks.