Tecelra and MAGE-A4 TCR Therapy: Engineering Safer, High-Affinity Adoptive T-Cell Immunotherapy
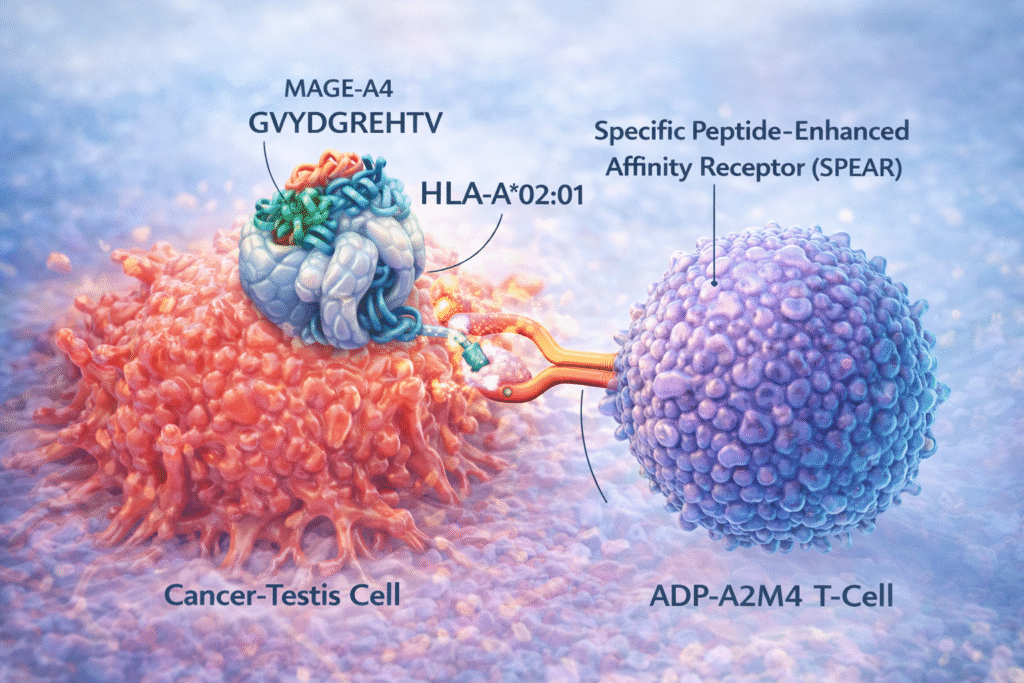
Adoptive (T-cell) TCR therapy utilizes the immune system’s T-cells to eliminate the cancer-causing cells from the body. Some of the primary approaches followed involves isolating the cancer targeting immune cells and allow their proliferation whereas others involve genetically engineering the immune cells, termed as TCR gene therapy, to enhance their cancer-fighting capabilities. Although T-cells are engineered to target the cancer cells, they display weak TCR cross-reactivity towards the normal tissue antigens, potentially leading to clinical toxicities.
The study by Sanderson and colleagues at the (Adaptimmune (ADAPY) tackled this problem head-on by asking a difficult question if a high-affinity TCR can be engineer that is both powerful and safe. ADP-A2M4 (named as Afamitresgene autoleucel, commonly labelled as Tecelra), is an affinity-enhanced TCR that recognizes a peptide from MAGE-A4 (GVYDGREHTV) expressed as a cancer-testis antigen by one of the most common HLA alleles (HLA-A*02:01). Supported by a comprehensive preclinical evaluation, the study demonstrates that ADP-A2M4 is presented as a personalized T-cell immunotherapy (SPEAR T-cell therapy) that specifically target the MAGE-A4 expressing cancer cells of different origins without major off-target cross-reactivity. In addition, to the findings of the ADP-A2M4 efficacy and specificity, the study highlights the framework it establishes for evaluating affinity-enhanced TCRs.
Study Outcomes:
- In vitro cell ELISA and ELISpot assay demonstrated strong cytokine IFNγ responses by the ADP-A2M4 in the presence of MAGE-A4 expressing A375 cell lines melanoma lots.
- ADP-A2M4 demonstrated effective cytotoxicity by killing HLA-A*02 and MAGE-A4-expressing Colo205 cell lines, in standard adherent cell culture.
- In vivo findings displayed dose-dependent anti-tumor effects against both single, large subcutaneous tumors and the small diffuse tumors, achieving the survival rate of 100%.
- ADP-A2M4 demonstrated no cross-reactivity to any of the iPSC-derived cell-subtypes like cardiomyocytes, astrocytes, endothelial cells, assessed by in vitro cytotoxic activity and IFNγ and GzB release.
- Although specific, ADP-A2M4 displays alloreactivity toward HLA-A*02:05, as assessed after screening of EBV-trans formed B-lymphoblastic cell lines expressing a wide range of HLA alleles using IFNγ cell-ELISA.
- ADP-A2M4 also displays cross-reactivity to MAGE-A8 and MAGE-B2, in the NALM6 cells (HLA-A*02:01+MAGE− cells transduced with these peptides, albeit weaker than those toward MAGE-A4+ tumor lines.
Future Applications:
The outcomes of this study paved the way for further clinical trials (NCT03132922, NCT04044768), where efficacy of TCR is being investigated in the urinary bladder, melanoma, head and neck, ovarian, non-small cell lung, esophageal, gastric, synovial sarcoma, or myxoid/round call liposarcoma. Tecelra received FDA accelerated approval for metastatic synovial sarcoma (commonly termed as Stage IV synovial sarcoma) in August 2024. This milestone provides broader clinical validation of TCR therapy like ADP-A2M4 and ADP-A2M4CD8 in ongoing clinical programs, for solid tumors. The approval builds increasing regulatory confidence in TCR platforms targeting MAGE-A4 antigens.